A new paper has identified an important property of rare variants: they tend to be population-specific: rare variants in Europeans are mostly not the same as rare variants in Asians or Africans. A corollary of this observation is that a sample of X global individuals is effectively much smaller than X for the purposes of identifying rare variants: you expect 1 in 1,000 individuals to possess an allele with 0.1% frequency, but if the allele occurs at 0.1% frequency in 250 Europeans, and at 0% frequency in 750 non-Europeans, then you expect to find 0.25 individuals in such a sample, in other words, you are very likely to miss it entirely.
The practical consequence of this, is to identify most rare variants in humans you need even larger samples: the 1000 genomes project is likely to miss many of them, and, correspondingly, a large part of the potential contribution of rare variants to disease. A second consequence is that if the so-called missing heritability is hiding in rare variants, different populations will get the same diseases but for different genomic reasons.
Was this unexpected?
This finding was not entirely unexpected. Consider the human species prior to its separation: it would harbor alleles ranging from very low to very high frequencies.
The very low-frequency alleles would have a high probability of being lost by drift in both the entire species (hence becoming irrelevant), or by founder effect in most human sub-populations. Only those that had some selective advantage might be expected to rise in frequency. Consequently, low-frequency alleles prior to the separation of Homo sapiens into regional sub-populations would mostly not persevere as low-frequency alleles today.
How about very low frequency alleles that arose after the separation of modern humans into Africans, Asians, Europeans, etc.? Again, the odds are stacked against those moving around: with a low migration rate between different human groups, it is fairly unlikely that a rare allele would migrate. Consequently, rare alleles that arose in regional populations were either lost by drift, or they grew in frequency (becoming non-rare), and, if, they persevered as rare variants, it is unlikely that they would be exchanged with other regional populations.
So, where do the rare variants mostly come from? They have to be mostly recent variants that have not had the time to either go extinct due, or to become common by drift, and their rarity and young age makes it unlikely that they had the chance to emigrate to other regional populations.
Demographic estimates
The paper's other major contribution is in providing some fairly tight estimates of human demographic parameters, much better than previous ones. A summary can be seen on the left. What I especially like about this is that the authors explicitly tackle the very young age for the European-East Asian split, without invoking a "long hiatus" as previous papers did. Here is their explanation:
The narrow confidence intervals on some of the parameters should not obscure the fact that the parameter estimates are model-dependent. As a simple example, a model that does not allow for migration would require more recent split times to produce similar levels of population divergence. The demographic history of the four populations considered is much more eventful than what is accounted for by our model. Additional geographically intermediate populations from the Near East and Central Asia that were not included in our analysis might contribute significantly to the allele frequency distribution as ghost populations (19).Incorporating an appropriate number of source populations for estimates of migration has been a general limitation of two-and three-population models under isolation migration coalescent, approximate Bayesian computation, and diffusion-based approaches. This limitation might explain why our estimate of the divergence between East Asians and Europeans is more recent than estimates based on archaeological evidence (18), but is comparable with estimates of 23 kya (20) under an approximate Bayesian computation approach and 25 kya under an isolation migration approach with mtDNA X and Y sequence data (21).
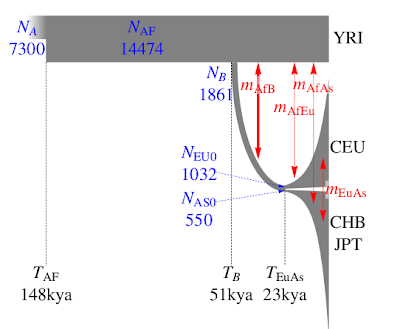
This makes excellent sense: we don't need to hypothesize that Proto-Eurasians spent tens of thousands of years in the Near East in a long stasis for no apparent reason; the archaeological record shows the arrival of anatomically modern humans in Europe long before 23,000 years ago. Indeed the earliest Europeans, and certainly those of 23,000 years ago already had typical Caucasoid physical characteristics. Hence, the idea that Europeans' ancestors were still marooned as undifferentiated Eurasians together with the ancestors of the Chinese difficult to swallow.
Instead, we can accept that East-West Eurasian differentiation began already shortly after Out-of-Africa, and the younger apparent divergence of Europeans and East Asians is due to gene flow, perhaps from an unsampled population that contributed genes to both.
Another interesting observation in the paper is that the divergence between Yoruba and Eurasians is a likely under-estimate of the Out-of-Africa event. This makes excellent sense, as West Africans are not expected to be identical with Proto-Eurasians or their closest African relatives. Hence, the 51ky is an over-estimate, and the true age seems to correspond well with the appearance of fully modern (in both appearance and behavior) Upper Paleolithic people in Eurasia, in full agreement with the recent scenario I posted on the blog, the end of Marine Isotope Stage 3, and the coalescence of most modern human Y-chromosomes.
Indeed, even the age of the African population (148kya) seems to be consistent with the recent re-dating of Y-chromosome Adam to 142kya. It would be interesting to include some African hunter-gatherers in the future; unfortunately, one of the great defects of the 1000 Genomes Project is its lack of African hunter-gatherers, which are, perhaps the most interesting populations for human origins research.
In any case, it is refreshing to see a genetics paper that tries to show how the genetic dates may deviate from the archaeological ones for good internal reasons, and seeks to explain the small discrepancies between the two by the limitations of genetic methods. This is much better than proposing archaeological implausible scenaria to "save" the genetic evidence, which, until this paper came along, did not really have the fairly tight confidence intervals necessary for genetic-archaeological correlations.
PNAS doi: 10.1073/pnas.1019276108
Demographic history and rare allele sharing among human populations
Simon Gravel et al.
High-throughput sequencing technology enables population-level surveys of human genomic variation. Here, we examine the joint allele frequency distributions across continental human populations and present an approach for combining complementary aspects of whole-genome, low-coverage data and targeted high-coverage data. We apply this approach to data generated by the pilot phase of the Thousand Genomes Project, including whole-genome 2–4× coverage data for 179 samples from HapMap European, Asian, and African panels as well as high-coverage target sequencing of the exons of 800 genes from 697 individuals in seven populations. We use the site frequency spectra obtained from these data to infer demographic parameters for an Out-of-Africa model for populations of African, European, and Asian descent and to predict, by a jackknife-based approach, the amount of genetic diversity that will be discovered as sample sizes are increased. We predict that the number of discovered nonsynonymous coding variants will reach 100,000 in each population after ∼1,000 sequenced chromosomes per population, whereas ∼2,500 chromosomes will be needed for the same number of synonymous variants. Beyond this point, the number of segregating sites in the European and Asian panel populations is expected to overcome that of the African panel because of faster recent population growth. Overall, we find that the majority of human genomic variable sites are rare and exhibit little sharing among diverged populations. Our results emphasize that replication of disease association for specific rare genetic variants across diverged populations must overcome both reduced statistical power because of rarity and higher population divergence.
Link

15 comments:
''unfortunately, one of the great defects of the 1000 Genomes Project is its lack of African hunter-gatherers, which are, perhaps the most interesting populations for human origins research.''
I agree, but judging from this statement on the informed consent background document of the 1000 genomes project it is unlikely that they would include these groups.
''The 1000 Genomes Project will not include samples from populations or communities
that are determined to be particularly vulnerable. For example, small, isolated
populations may make participants vulnerable to identification, stigmatization, or
invasion of privacy. Other populations may be vulnerable due to factors such as
socioeconomic status. Determinations regarding vulnerability will be made by the
project Steering Committee in consultation with the Samples/ELSI Group and other
relevant experts.''
We understand that there were already ‘typical Caucasoid physical characteristics’ by 23,000 years ago, but what can you tell us about the physical characteristics of East Asians at that time?..
but what can you tell us about the physical characteristics of East Asians at that time?..
The earliest full-blown Mongoloids appear in Neolithic China, as far as I can determine
http://dienekes.blogspot.com/2005/08/earliest-examples-of-four-major-racial.html
Upper Cave people from China seem to have "Polynesioid" affinities, and people from southeast Asia "Australoid" affinities. In any case, they look different from Europeans, which is inconsistent with them diverging as recently as 23,000 years ago.
I would also add that Soviet anthropologists identified Mongoloid characteristics in Upper Paleolithic Siberians
http://dienekes.awardspace.com/blog/archives/000399.html
Also, Paleoindians (who migrated into the New World c. 15kya) do not yet possess a Mongoloid appearance, even though their present-day descendants are undeniably aligned morphologically with East Asians to a substantial degree.
In general, the emergence of Mongoloids is more obscure than that of the Caucasoids of West Eurasia.
"Also, Paleoindians (who migrated into the New World c. 15kya) do not yet possess a Mongoloid appearance, even though their present-day descendants are undeniably aligned morphologically with East Asians to a substantial degree."
There's no evidence for a migration to the New World 15,000 years. The earliest skulls with a generalized Mongoloid shape appear in North America and not in Asia: "Peter Brown (1999) of the Department of Anthropology and Paleoanthropology at the University of New England evaluated three sites with early East Asian modern human skeletal remains (Liujiang, Liuzhou, Guangxi, China; Shandingdong Man of (but not Peking Man) Zhoukoudian's Upper Cave; and Minatogawa in Okinawa) dated to between 10,175 to 33,200 years ago, and finds lack of support for the conventional designation of skeletons from this period as "Proto-Mongoloid"; this would make Neolithic sites 5500 to 7000 years ago (e.g. Banpo) the oldest known Mongoloid remains in East Asia, younger than some in the Americas. He concludes that the origin of the Mongoloid phenotype remains unknown, and could even lie in the New World" (http://en.wikipedia.org/wiki/Mongoloid_race).
This means, by the logic of the authors of this paper and by Dienekes's logic, that the ostensibly recent separation of East Asians and Amerindians can be explained as an artifact of gene flow from America to Asia some 12,000 years ago. That population may have carried with it back to the Old World the incipient "Mongoloid" morphology. Some aspects of this morphology, however, may be plesiomorphic, as shovel-shaped incisors (90% in Amerindians, 85% in Northern Mongoloids) are found at high frequencies in Neandertals and at low frequencies in Africans.
Ok, you've said your piece; let me remind you that the Out-of-America theory of human origins is not allowed in this blog.
Dieneke, what modern human races/subspecies were there in various regions of the world prior to the emergence of the extant modern human races/subspecies and how different were they from each other and from the extant modern human races/subspecies?
I'm just stating facts and providing references. Just like you...
Thank you for the information Dienekes, very interesting read. I forgot to ask about Sub-Saharan Africans. What can you tell us about their physical characteristics 23,000 years ago such as was it negroid, etc?
"Instead, we can accept that East-West Eurasian differentiation began already shortly after Out-of-Africa, and the younger apparent divergence of Europeans and East Asians is due to gene flow, perhaps from an unsampled population that contributed genes to both."
Seems to me that India/Pakistan would be a good place for that. During cold/dry times, with the monsoon shut off, it would have supported an East/West separation, since much of the center of the region was inhospitable desert. During times the monsoon switched on again, the area supported mixing and also quick population growth that would have wanted to migrate outwards.
These guys used a mutation rate of 2.36 x 10-8 per generation, a rate determined from the measured difference between humans and chimps
and an estimate of the human/chimp split time based upon fossil evidence. Recent direct measurements on family triads indicate the that the mutation rate is approximately 1.1 x 10-8 per generation: if correct, this would give substantially different split times.
Much older.
If 1.1x10-8 is more accurate, then that would push African-Eurasian divergence to 109ky, which does correspond to a known event (the appearance of modern humans in the Levant, where previously there lived Neandertals).
It would also push European-East Asian divergence to 50ky, which seems earlier than the appearance of modern humans in Europe, but could be saved, I guess, if either the divergence began somewhere in Asia, before the arrival of AMH in Europe and/or there is a little archaic admixture in East Asians inflating the divergence times; archaic admixture in Europeans seems less likely, because the only candidate is Neandertals, and we already know that Europeans have no more Neandertal genes than East Asians do.
"Dieneke, what modern human races/subspecies were there in various regions of the world prior to the emergence of the extant modern human races/subspecies and how different were they from each other and from the extant modern human races/subspecies?"
I would expect, with the presumed lesser mobility of pre-modern species, the diversity of such species would be greater than that of the modern human species. However I have never seen any analysis of such diversity, excepting in a book by Lee Berger claiming differences between northern and southern Australopithecus species.
"I would also add that Soviet anthropologists identified Mongoloid characteristics in Upper Paleolithic Siberians"
It would make complete sense that the Mongoloid phenotype developed there. Most characteristics associated with East Asians appear to be adaptations to a cold, highly reflective environment.
I would expect, with the presumed lesser mobility of pre-modern species, the diversity of such species would be greater than that of the modern human species. However I have never seen any analysis of such diversity, excepting in a book by Lee Berger claiming differences between northern and southern Australopithecus species.
My question was exclusively about modern human subspecies (I prefer the word subspecies to the word race, as it is clearer in meaning), whether extant or non-extant; archaic (=pre-modern) Homo and pre-Homo hominids is another issue.
"I forgot to ask about Sub-Saharan Africans. What can you tell us about their physical characteristics 23,000 years ago such as was it negroid, etc?"
According to the present data, the Negroid type could be quite recent in Africa. The oldest is the 11 kyr Iwo Eleru skullcap from Nigeria and the 6.4 kyr skeleton from Asselar, eastern Mali. The Khoisanid type is also likely of Holocene age: see "Early to mid-Holocene South African Later Stone Age human crania exhibit a distinctly Khoesan morphological pattern," by D.D. Styndera, R.R. Ackermanna and J.C. Sealy.
On the other hand, the earliest South African skull, namely the 36,000 year old Hofmeyr, clustered with Upper Paleolithic Europeans. See F. E. Grine, FE; R. M. Bailey, K. Harvati, R. P. Nathan, A. G. Morris, G. M. Henderson, I. Ribot, A. W. G. Pike (12 January 2007). "Late Pleistocene Human Skull from Hofmeyr, South Africa, and Modern Human Origins". Science 315 (5809): 226–229.
So, Dienekes's Upper Paleolithic Caucasoids may have been in South Africa before the formation of modern Khoisans and Negroids.
"It would also push European-East Asian divergence to 50ky, which seems earlier than the appearance of modern humans in Europe, but could be saved, I guess, if ... the divergence began somewhere in Asia, before the arrival of AMH in Europe "
I think there is little question that Europe initially was (mostly) populated from Pakistan/India rather than the Levant. At the documented around ~40ky ago, that is not so different from 50k if we allow for some traveling time from ~Pakistan via Neanderthal- "infested" regions to Europe - probably using a northern corridor first, before a southern one once it became safe, and accounting for some cold/dry separation time within that sub-continent.
Post a Comment